(Science Magazine=E. Lehman et al.) 10 Jan 2020
Summary
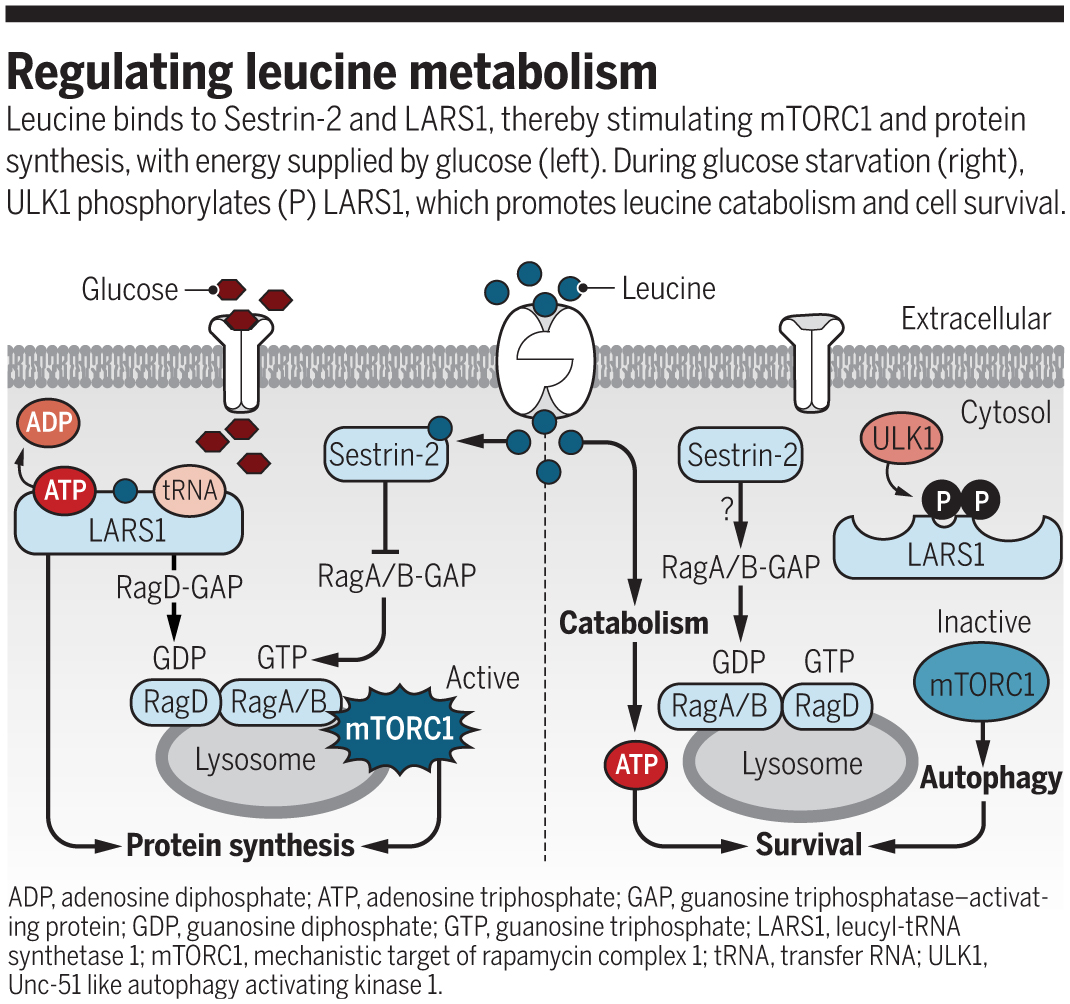
Mammalian cell growth is tightly linked to adequate supplies of growth factors and nutrients, including glucose and amino acids. The mechanistic target of rapamycin complex 1 (mTORC1) functions as a coincidence detector that supports anabolic metabolism when convergent growth factor– and nutrient-derived signals trigger mTORC1 kinase activation. Conversely, nutrient starvation suppresses mTORC1 activity and triggers a shift to catabolic pathways, such as autophagy, to support cell survival under austere conditions. An elaborate signaling apparatus has evolved to harmonize mTORC1 kinase activation and protein synthesis with supplies of leucine and other amino acids (1). Earlier findings implicated the leucyl-transfer RNA (tRNA) synthetase 1 (LARS1) as a proximate sensor of leucine availability (2). On page 205 of this issue, Yoon et al. (3) report that glucose modulates the functions of LARS1 in leucine sensing and disposition, thereby coordinating leucine-dependent mTORC1 activation and protein synthesis with cellular energy stores.
http://www.sciencemag.org/about/science-licenses-journal-article-reuse
This is an article distributed under the terms of the Science Journals Default License.
Original Article Link : https://science.sciencemag.org/content/367/6474/146
